ABOUT US
HKU EARTH SCIENCES

Our department was established in 1995 to meet local demand for Earth science expertise. This is because as a densely populated city, Hong Kong’s subtropical location, local geology and limited water and mineral resources present significant societal challenges. We are proud of these origins and continue to help improve our home through our research, teaching and graduates.

Learn More
RESEARCH GROUPS
To engage in research on global, regional and local geological problems, we use a team approach. We operate three research groups: Solid Earth and Planet, Earth History and Global Change & Applied/Urban Geosciences

Learn More
RECENT PUBLICATIONS

Ambient noise differential adjoint tomography reveals fluid-bearing rocks near active faults in Los Angeles
Xin Liu, Gregory C. Beroza & Hongyi Li
Water scarcity is a pressing issue in California. We develop ambient noise differential adjoint tomography that improves the sensitivity to fluid-bearing rocks by canceling bias caused by noise sources. Here we image the shallow S-wave velocity structure using this method beneath a linear seismic array (LASSIE) in Los Angeles Basin, which shows significant velocity reduction marking a major regional water producer, the Silverado aquifer, along with other fluid-bearing structures. Based on the S-wave tomography and previous P-wave studies, we derive the porosity in Long Beach and discover that the rock from 1-2 km depth surrounding the Newport-Inglewood Fault contains abundant fluids with pore-fluid fraction ~0.33. The high-porosity rock around the fault coincides with previously observed week-long shallow seismicity south of LASSIE array in Long Beach. The imaged S-wave velocity in the top layer shows a similar trend in the geotechnical layer Vs 30, suggesting additional applications to ground motion prediction.

Click Here
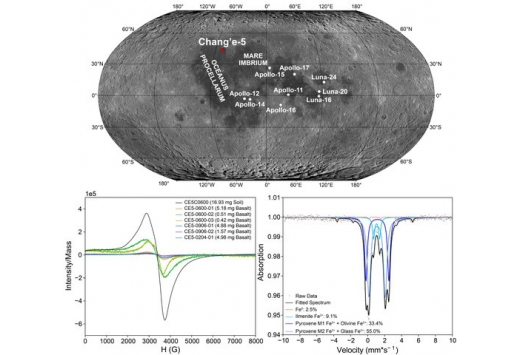
First magnetic and spectroscopic constraints on attenuated space weathering at the Chang’e-5 landing site
Yuqi Qian, Long Xiao, James W. Head, Joseph Michalski
Dr. Yuqi Qian and Dr. Joseph Michalski from the Department of Earth Sciences, The University of Hong Kong, has publised a paper in Icarus on Nov. 24, 2023, documenting the space weathering effects of the newly returned Chang’e-5 lunar soils. In this paper, they conducted the first vibrating sample magnetometer measurement, the first electron paramagnetic resonance spectrometer measurement, and the first Mössbauer spectrometer measurement of Chang’e-5 soils. They found that Chang’e-5 lunar soil is among the most immature returned lunar soils from a never-sampled young geological unit.

Click Here
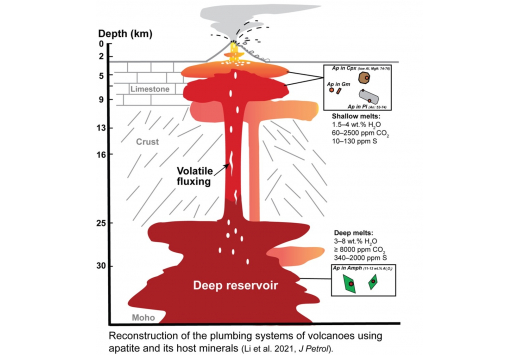
Apatite crystals reveal melt volatile budgets and magma storage depths at Merapi volcano, Indonesia. Journal of Petrology egaa100.
Li, W.*, Costa, F. & Nagashima, K. (2021)
Merapi Volcano in central Java (Indonesia) is one of the most active volcanoes in the world, and it had about 27 eruptions in the last century (e.g. Voight et al., 2000). Most eruptions at Merapi are dome-forming events with VEIs (Volcanic Explosivity Index; Newhall & Self, 1982) between 1–3, such as that occurred in 2006 (Ratdomopurbo et al., 2013). But in 2010, Merapi produced an unexpectedly explosive eruption (VEI 4), which turned out to be the largest eruption in the last hundred years and caused severe hazards to the local community (Surono et al., 2012). Although the eruptions in 2006 and 2010 have very different eruptive styles, most of the materials emitted from the two eruptions (except some from the very last eruptive stage in 2010; Komorowski et al., 2013) show similar petrological and geochemical features, i.e. similar compositions of the bulk rocks (as basaltic andesite), mineral assemblages (see below) and phenocryst compositions (Costa et al., 2013; Preece, 2014; Preece et al., 2014; Erdmann et al., 2016; Preece et al., 2016). A critical question is what caused the larger and more hazardous eruption in 2010, compared to the 2006 eruption and earlier events, and in relation to this we provide below a brief review of the observations from the literature.

Click Here
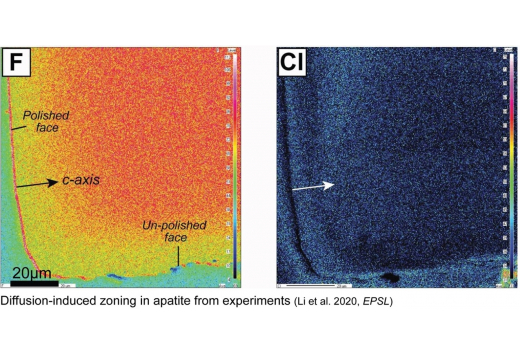
Multicomponent diffusion of F, Cl, OH in apatite with application to magma ascent rates. Earth and Planetary Science Letters
Li, W.*, Chakraborty, S., Nagashima, K., & Costa, F. (2020)
1)Volatile element zoning in apatite can be used to reveal the timescales of magma ascent. 2)Multicomponent diffusion of F-Cl-OH, and anisotropic diffusion of Cl and OH in apatite. 3)Zoned groundmass apatite reflects magma ascent times of days before eruption. 4)An online tool for apatite diffusion modelling and estimating magma ascent times.

Click Here

Petrogenesis and tectonic setting of Neoarchean tonalitic-trondhjemitic-granodioritic gneisses in the Xiwulanbulang area of the Yinshan block, North China craton. Geological Society of America Bulletin
Wang X, Zhang J, Liu Q
As one of the major Archean microcontinental blocks in the North China craton, the Yinshan block consists of the Guyang granite-greenstone belt and the Wuchuan high-grade complex, both of which preserve large volumes of Neoarchean tonalitic-trondhjemitic-granodioritic (TTG) gneisses

Click Here

Early Prosperity of Iron Bacteria at the End of the Paleoproterozoic Era
Qiu, Y., Qin, L., Huang, F., Zhao, T., & Li, Y. (2022).
Various iron formation (IFs) document the interplay between the geosphere and the early biosphere during their co-evolution. At the transition between the Paleo- and Mesoproterozoic eras, the oxidation of the atmosphere and ocean resulted in the waning of the typical marine deposited banded IFs. Here, we report the ∼1.7 Ga Yunmengshan IF at the southern margin of the North China Craton deposited in the intertidal to upper subtidal zone belonging to river delta of coastal environments. The combined REE + Y patterns and negative δ56Fe with positive δ53Cr values reveal terrestrial precipitation of iron oxide and an enhanced continental oxidative weathering at the end of Paleoproterozoic era due to increased atmospheric oxygen level. The oxygen level happened to have facilitated the early prosperity of microaerophilic Fe(II)-oxidation bacteria on Earth's terrestrial surface, which is recorded by the coastal shallow water iron deposits globally characterized by their small scale and subaerial features.

Click Here

Photoferrotrophic Bacteria Initiated Plate Tectonics in the Neoarchean
Zhang, S., Li, Y., Leng, W., & Gurnis, M. (2023)
Plate tectonics distinguishes Earth from the other terrestrial planets but its initiation mechanism and onset time are debated. We propose plate tectonics was initiated by the deposition of magnetite-rich banded iron formations (BIFs) through biogeochemical iron cycling in Neoarchean oceans. In the photic zone of proto-continental margins, photoferrotrophic bacteria efficiently oxidized the dissolved Fe(II) and induced massive precipitation of ferric oxyhydroxide, which would rapidly react with Fe(II)-rich hydrothermal fluids from coeval vigorous volcanism in Neoarchean oceans to produce magnetite-rich BIFs. Mechanical models demonstrate that the localization of high-density BIF deposition near proto-continents induces collapse of the lithosphere and can initiate the earliest subduction. The peak deposition of BIFs in 2.75–2.40 Ga provides a time constraint on the inception of plate tectonics.

Click Here
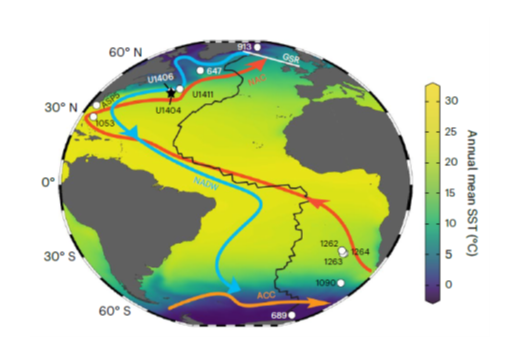
Oxygenated deep waters fed early Atlantic overturning circulation upon Antarctic glaciation
Wang, H., Liu, W., Lu, H., Zhang, Y., Liang, Y., He, Y., Bohaty, S. M., Wilson, P. A. & Liu, Z.
: We used a novel method to trace oxygenation using microbial biomarkers to infer the history of early Atlantic Meridional Overturning Circulation (AMOC). Our results suggest that AMOC-feed waters became better oxygenated upon initiation of Antarctic glaciation at the Eocene/ Oligocene transition, ~34 Ma. Our findings indicate a close association between the inception of Antarctic glaciation and AMOC and suggest that both vertical mixing and wind-driven upwelling in the Southern Ocean were key to fully establishing AMOC as an agent of deep-ocean ventilation.

Click Here

Spatial patterns of Holocene temperature changes over mid-latitude Eurasia
Jiang, J. Meng, B., Wang, H., Liu, H., Song, M., He, Y., Zhao, C., Chu, G., Krivonogov, S., Liu, W. & Liu, Z.
We have found contrasting Holocene temperature trends over mid-latitude Eurasia, with strong implications for the Holocene temperature conundrum, highlighting that spatial patterns of Holocene temperature changes should be re-considered in both record integrations and model simulations.

Click Here

NEWS

Final Year Student received second runner-up prize in the AGS (HK) Final Year Projects Competition 2023
2023.12.22
PhD student Zihan Liu won the championship in Young Persons Lecture Competition (Hong Kong heat) 2023.
2023.08.28

Geologists Team at HKU Becomes First in Hong Kong to Retrieve Lunar Samples - Set to Unveil the Mysteries of the Moon's Volcanic History
2023.08.07

Martian ancient calderas – exploration of under-recognized early Martian volcanism
2023.01.17

HKU Stephen Hui Geological Museum Curating an Exhibition on Poland’s Distinctive Landforms In Celebration of the International Geodiversity Day
2022.10.07

HKU geologist proposes the number of ancient martian lakes might have been dramatically underestimated by scientists
2022.10.07

Final Year Student received first runner-up prize in the AGS (HK) Final Year Projects Competition 2022
2022.08.02

Equipment booking system will be down on June 1 -3
2021.05.28

Final Year Student received second runner-up prize in the AGS (HK) Final Year Projects Competition 2023
2023.12.22

PhD student Zihan Liu won the championship in Young Persons Lecture Competition (Hong Kong heat) 2023.
2023.08.28

Geologists Team at HKU Becomes First in Hong Kong to Retrieve Lunar Samples - Set to Unveil the Mysteries of the Moon's Volcanic History
2023.08.07

Martian ancient calderas – exploration of under-recognized early Martian volcanism
2023.01.17

HKU Stephen Hui Geological Museum Curating an Exhibition on Poland’s Distinctive Landforms In Celebration of the International Geodiversity Day
2022.10.07

HKU geologist proposes the number of ancient martian lakes might have been dramatically underestimated by scientists
2022.10.07

Final Year Student received first runner-up prize in the AGS (HK) Final Year Projects Competition 2022
2022.08.02

Equipment booking system will be down on June 1 -3
2021.05.28

Final Year Student received second runner-up prize in the AGS (HK) Final Year Projects Competition 2023
2023.12.22

PhD student Zihan Liu won the championship in Young Persons Lecture Competition (Hong Kong heat) 2023.
2023.08.28

Geologists Team at HKU Becomes First in Hong Kong to Retrieve Lunar Samples - Set to Unveil the Mysteries of the Moon's Volcanic History
2023.08.07

Martian ancient calderas – exploration of under-recognized early Martian volcanism
2023.01.17

HKU Stephen Hui Geological Museum Curating an Exhibition on Poland’s Distinctive Landforms In Celebration of the International Geodiversity Day
2022.10.07

HKU geologist proposes the number of ancient martian lakes might have been dramatically underestimated by scientists
2022.10.07

Final Year Student received first runner-up prize in the AGS (HK) Final Year Projects Competition 2022
2022.08.02

Equipment booking system will be down on June 1 -3
2021.05.28

Final Year Student received second runner-up prize in the AGS (HK) Final Year Projects Competition 2023
2023.12.22

PhD student Zihan Liu won the championship in Young Persons Lecture Competition (Hong Kong heat) 2023.
2023.08.28

Geologists Team at HKU Becomes First in Hong Kong to Retrieve Lunar Samples - Set to Unveil the Mysteries of the Moon's Volcanic History
2023.08.07

Martian ancient calderas – exploration of under-recognized early Martian volcanism
2023.01.17

HKU Stephen Hui Geological Museum Curating an Exhibition on Poland’s Distinctive Landforms In Celebration of the International Geodiversity Day
2022.10.07

HKU geologist proposes the number of ancient martian lakes might have been dramatically underestimated by scientists
2022.10.07

Final Year Student received first runner-up prize in the AGS (HK) Final Year Projects Competition 2022
2022.08.02

Equipment booking system will be down on June 1 -3
2021.05.28

Final Year Student received second runner-up prize in the AGS (HK) Final Year Projects Competition 2023
2023.12.22

PhD student Zihan Liu won the championship in Young Persons Lecture Competition (Hong Kong heat) 2023.
2023.08.28

Geologists Team at HKU Becomes First in Hong Kong to Retrieve Lunar Samples - Set to Unveil the Mysteries of the Moon's Volcanic History
2023.08.07

Martian ancient calderas – exploration of under-recognized early Martian volcanism
2023.01.17

HKU Stephen Hui Geological Museum Curating an Exhibition on Poland’s Distinctive Landforms In Celebration of the International Geodiversity Day
2022.10.07

HKU geologist proposes the number of ancient martian lakes might have been dramatically underestimated by scientists
2022.10.07

Final Year Student received first runner-up prize in the AGS (HK) Final Year Projects Competition 2022
2022.08.02

Equipment booking system will be down on June 1 -3
2021.05.28

Final Year Student received second runner-up prize in the AGS (HK) Final Year Projects Competition 2023
2023.12.22

PhD student Zihan Liu won the championship in Young Persons Lecture Competition (Hong Kong heat) 2023.
2023.08.28

Geologists Team at HKU Becomes First in Hong Kong to Retrieve Lunar Samples - Set to Unveil the Mysteries of the Moon's Volcanic History
2023.08.07

Martian ancient calderas – exploration of under-recognized early Martian volcanism
2023.01.17

HKU Stephen Hui Geological Museum Curating an Exhibition on Poland’s Distinctive Landforms In Celebration of the International Geodiversity Day
2022.10.07

HKU geologist proposes the number of ancient martian lakes might have been dramatically underestimated by scientists
2022.10.07

Final Year Student received first runner-up prize in the AGS (HK) Final Year Projects Competition 2022
2022.08.02

Equipment booking system will be down on June 1 -3
2021.05.28

Final Year Student received second runner-up prize in the AGS (HK) Final Year Projects Competition 2023
2023.12.22

PhD student Zihan Liu won the championship in Young Persons Lecture Competition (Hong Kong heat) 2023.
2023.08.28

Geologists Team at HKU Becomes First in Hong Kong to Retrieve Lunar Samples - Set to Unveil the Mysteries of the Moon's Volcanic History
2023.08.07

Martian ancient calderas – exploration of under-recognized early Martian volcanism
2023.01.17

HKU Stephen Hui Geological Museum Curating an Exhibition on Poland’s Distinctive Landforms In Celebration of the International Geodiversity Day
2022.10.07

HKU geologist proposes the number of ancient martian lakes might have been dramatically underestimated by scientists
2022.10.07

Final Year Student received first runner-up prize in the AGS (HK) Final Year Projects Competition 2022
2022.08.02

Equipment booking system will be down on June 1 -3
2021.05.28

Final Year Student received second runner-up prize in the AGS (HK) Final Year Projects Competition 2023
2023.12.22

PhD student Zihan Liu won the championship in Young Persons Lecture Competition (Hong Kong heat) 2023.
2023.08.28

Geologists Team at HKU Becomes First in Hong Kong to Retrieve Lunar Samples - Set to Unveil the Mysteries of the Moon's Volcanic History
2023.08.07

Martian ancient calderas – exploration of under-recognized early Martian volcanism
2023.01.17

HKU Stephen Hui Geological Museum Curating an Exhibition on Poland’s Distinctive Landforms In Celebration of the International Geodiversity Day
2022.10.07

HKU geologist proposes the number of ancient martian lakes might have been dramatically underestimated by scientists
2022.10.07

Final Year Student received first runner-up prize in the AGS (HK) Final Year Projects Competition 2022
2022.08.02

Equipment booking system will be down on June 1 -3
2021.05.28

Final Year Student received second runner-up prize in the AGS (HK) Final Year Projects Competition 2023
2023.12.22

PhD student Zihan Liu won the championship in Young Persons Lecture Competition (Hong Kong heat) 2023.
2023.08.28

Geologists Team at HKU Becomes First in Hong Kong to Retrieve Lunar Samples - Set to Unveil the Mysteries of the Moon's Volcanic History
2023.08.07

Martian ancient calderas – exploration of under-recognized early Martian volcanism
2023.01.17

HKU Stephen Hui Geological Museum Curating an Exhibition on Poland’s Distinctive Landforms In Celebration of the International Geodiversity Day
2022.10.07

HKU geologist proposes the number of ancient martian lakes might have been dramatically underestimated by scientists
2022.10.07

Final Year Student received first runner-up prize in the AGS (HK) Final Year Projects Competition 2022
2022.08.02

Equipment booking system will be down on June 1 -3
2021.05.28

EVENTS

2024.04.16
Miss LIU Xiaoyu (Supervisor: Prof . WR Li)
Department of Earth Sciences, University of Hong Kong
Machine Learning Geothermobarometry using Clinopyroxene

2024.04.16
Mr. SHANG Kun (Supervisor: Prof. GC Zhao)
Department of Earth Sciences, University of Hong Kong
Surface-derived materials in TTG and Komatiite? hint from mass-independent fractionation of sulfur isotopes

2024.02.27
Mr. SU Jintao (Primary Supervisor: Dr. LUO Xin)
Department of Earth Sciences, HKU
Resilience and Weathering Regimes of Groundwater System under Changing Climate and Human Perturbation

2024.02.27
Miss QIN Yonghui (Supervisor: Prof. Ryan McKenzie)
Department of Earth Sciences, HKU
Organic Carbon Sequestration in Coastal Environments

2024.02.21
Dr. Lihui Chai
Institute of Geology and Geophysics, Chinese Academy of Sciences
Research of Mercury’s Magnetosphere Using a Global Three-Dimensional Hybrid Model

2024.02.20
Mr. LUO Hongyang (Primary Supervisor: Prof. B.Z. Zhang)
Department of Earth Sciences, University of Hong Kong
Global Planetary Magnetosphere Simulation: Model Development and Application

2024.02.20
Mr. WANG Zhengyang (Supervisor: Prof. B.Z. Zhang)
Department of Earth Sciences, University of Hong Kong
The affection of internal mechanism on Jupiter’s aurora and its magnetosphere

2024.02.06
Miss HU Yufan
Department of Earth Sciences, University of Hong Kong
Archaeological Field Work of Shakengnan, a Prehistoric Site at Jiesheng Peninsula, East Guangdong Province, China

2024.01.31
Ms. Wensi GUO (Supervisor: Prof. JJ Jiao, HKU; Prof. ZHENG Yan, SUSTech)
Department of Earth Sciences, HKU
Integrated Surface Water and Groundwater Modeling of the Baoding Plain in Northern China: Towards Groundwater Sustainability through Managed Aquifer Recharge

2024.01.30
Mr. Anyang DING (Primary Supervisor: Prof GC Zhao)
Department of Earth Sciences, HKU
Banded Iron Formation deposition through photoferrotrophy and the evolution of Earth surface chemistry and climate

2024.01.30
Miss XIA Yangyang (Supervisor: Prof CHANG Su-Chin)
Department of Earth Sciences, HKU
Fossil charcoal from the Middle-Upper Triassic of western Junggar Basin, Xinjiang, and its palaeoecological significance

2024.01.30
Mr. LIU Zhaojia (Supervisors: Prof. Nicole Khan & Prof. Celia Schunter)
Department of Earth Sciences, HKU
Exploring the potential of foraminiferal environmental DNA to reconstruct sea level

2024.01.29
Mr. LYU Tianyang (Supervisor: Prof MH Lee)
Department of Earth Sciences, HKU
Integrated Study of Lid Modes and Plate Tectonic Patterns in Planetary Evolution: Diversity, Commonalities, and Evolutionary Mechanisms

2024.01.29
Prof. Jie LIAO
School of Earth Sciences and Engineering, Sun Yat-Sen University
Will the South China Sea disappear through subduction?

2024.01.23
Mr. Zihan LIU (Supervisor: Prof. Louis NY Wong)
Department of Earth Sciences, HKU
Rock Strengthening Mechanisms Upon Heating: Experimental and Numerical Research

2024.01.23
Mr. Zhicheng WANG (Supervisor: Prof. J Michalski)
Department of Earth Sciences, HKU
Planetary mineralogy and geologic history of Mars

2024.01.16
Mr. Guodong CHEN
Department of Earth Sciences, HKU
Fracture Network Characterization and Geothermal Energy Design Optimization

2024.01.16
Mr. Giancarlo DEFRANCESCO
Department of Earth Sciences, HKU
Investigating ophiolite weathering via lithium isotopes in the Indo-Burma range of northeast India

2023.11.23
Mr. YIN Qianfeng (Supervisor: Dr. BZ Zhang)
Department of Earth Sciences, University of Hong Kong
To:Hazel Lam wing Shuet 22 Soudrey Way CF10 5FW Wales, UK

2023.11.21
Dr. Yuqi QIAN
Department of Earth Sciences and Laboratory for Space Research, The University of Hong Kong















